For last 50 years all Governments have been controlling human and industrial activities which can cause degradation to environment. Despite this, pressure of the environment has increased in last 10-20 years which has brought Paris Agreement. This agreement is legally binding agreement adopted by 196 nations at United Nations Climate Change conference (COP-21). The Agreement requires all nations to reach peak Greenhouse Gas emission by 2025 and decline by 43% by 2030. The aim of the agreement is to ensure limit the temperature rise to 1.5-degree above the pre-industrial level. To achieve the targets set by Paris Agreement it is accepted that multi-dimensional approach will be required. The target set will require people centric approach fully backed by regulatory framework, use of cost-effective technologies and sufficient finances.
Green House Gases
Greenhouse gas (GHG) emissions from human activities contributes to climate change. Carbon dioxide (CO2) produced due to burning of fossil fuels is a major contributor to the climate change. Other gases which are categorised as GHG are methane (CH4), nitrous oxide (N2O) and fluorinated gases. It is estimated that the CO2 emission in year 2023 was about 37.55 billion tonnes. This means average CO2 emission per person per year was about 5.52 tonnes for a population of 6.8 billion. This is much higher than the estimated rate of 2.3 tonnes per year per person required by Paris Agreement to ensure that increase in temperature does not exceed 1.5-degree C, over pre-industrial level.
Scientist working on to reduce the effect of GHG, have proposed two prong strategy. The reduction of emission should be accompanied by removal of CO2 from power and process plant chimneys. International Energy Agency (IEA, 2019) in its report titled ‘Putting use to use, creating value from emissions’, suggested following usages of CO2:
- CO2 storage
- CO2 derived fuels such as methane, methanol, gasoline, and aviation fuel.
- CO2 derived chemicals such as olefins, used for production of plastics and aromatics which are used for health and food processing.
- Building materials from minerals and CO2 such as replacing water with CO2 for concrete preparation.
- Building material from waste and CO2 such as iron slag and coal fly ash.
- Crop yield boosting with CO2.
Except for CO2 storage and utilisation (CCUS) all other have limited potential due to current development of technology.
Liquid fuels
Scientists have been conducting research to use CO2 to produce hydrogen and liquid fuels for almost two decades. The liquid fuel production process requires use renewable energy to split water into hydrogen and oxygen through electrolysis, and then combining the hydrogen with carbon dioxide to produce hydrocarbons through Fischer-Tropsch reaction. The feasibility of this approach depends on several factors:
Energy Input:
Splitting of water requires high energy and therefore the power should be sourced from renewable sources.
Technology:
All components of the process must have been tested at the pilot plant scale before any experimental plant can be constructed. The efficiency of the process is important due to high energy requirement for splitting of water. The production process should be environmentally friendly including production of energy and disposal of waste from the facility.
Economic Viability:
The technology must be assessed based on capital cost of the facilities and cost of operation. In final analysis, the cost of production of fuel must be same as the fuels in use.
Status of Research
Massachusetts Institute of Technology (MIT):
In 2023, MIT announced that they have developed a process that can convert carbon dioxide into formate, a liquid or solid material that can be used like hydrogen or methanol to power a fuel cell and generate electricity. The present work is at laboratory scale and MIT expects it to be scalable. Potassium or sodium formate is already being produced at industrial scales (used for de-icing of roads) and is nontoxic, non-flammable, easy to store and transport. The conversion efficiency was stated at 90%.
Stanford University:
In 2022, the University announced that they have developed a catalyst which can produce 1000 times more butane in laboratory trials. This catalyst can increase production of hydrocarbons in chemical reactions. The catalyst is composed of the elemental ruthenium, a rare metal.
University of Texas at Arlington (UTA):
In 2016, UTA had announced that the University has developed a one-step photo-thermo-chemical process which turns carbon dioxide and water into oxygen and liquid hydrocarbons. The reactor operating temperature was stated as 180-200 degree C and pressure up to six atmospheres. The process used titanium dioxide catalyst.
University of Cambridge:
In 2023, University of Cambridge announced that they have developed a laboratory scale device to convert CO2 and water into ethanol and n-propanol, in a single step using sunlight. The researchers have informed that they have used copper and palladium-based catalyst to optimise the process.
Infinium:
The company has announced that it has completed a commercial scale plant in March 2024 to produce fuel for heavy transport which can be used in existing internal combustion engines. The facility is using renewable energy, CO2, and water. Infinium claims that the technology has net zero impact on the environment. In addition to the above demonstration facility, the company has announced that a funding of USD 75 million has been secured from Bill Gate foundation for development of catalyst. Bill Gate foundation provides financial support to development projects.
However, the view that the technology is neutral is contested by the researchers in Columbia University. As per the University, the gas captured is only temporarily stored and when e-fuel is used, the CO2 will be released. Hence this is not neutral and there will some net addition to the environment.
Review:
The information provided above is shows that several organisations are working on various aspects of the technology development to produce liquid fuels such as methanol, diesel, and aviation fuels. Such fuels are good for transport as these are low carbon carriers. As claimed by Infinium, at least one demonstration plant has been completed and ready for commercial development.
Commercial scale facilities planned by Infinium.
Infinium have announced that they are planning commercial scale facilities in USA, EU, Japan, and Australia. These facilities will produce e-SAF (sustainable aviation fuel), e-Diesel and e-naphtha. These fuels have lower carbon carriers and produce lower NOx, SOx, and other toxins.
Corpus Christi, Texas, USA:
This is first commercial scale plant commissioned in October 2023 to produce 8300 litre per day of fuels for heavy transport. The fuel produced by the facility can be used for aviation, heavy transport, and industry.
The process scheme adopted by Infinium uses electrolysers to break-down water into hydrogen, and oxygen using electricity generated by wind and solar farms. Hydrogen produced is transported to reactor where it reacts with CO2 extracted from refinery stack gases. The plant is using patented catalyst. The fuel produced is compatible for use in aviation and heavy transport without change to internal combustion engines. Naphtha produced can be used by the industry. Process schematic is shown in Figure 1. Using this facility as the base, Infinium plans to develop 13 projects all over the world.
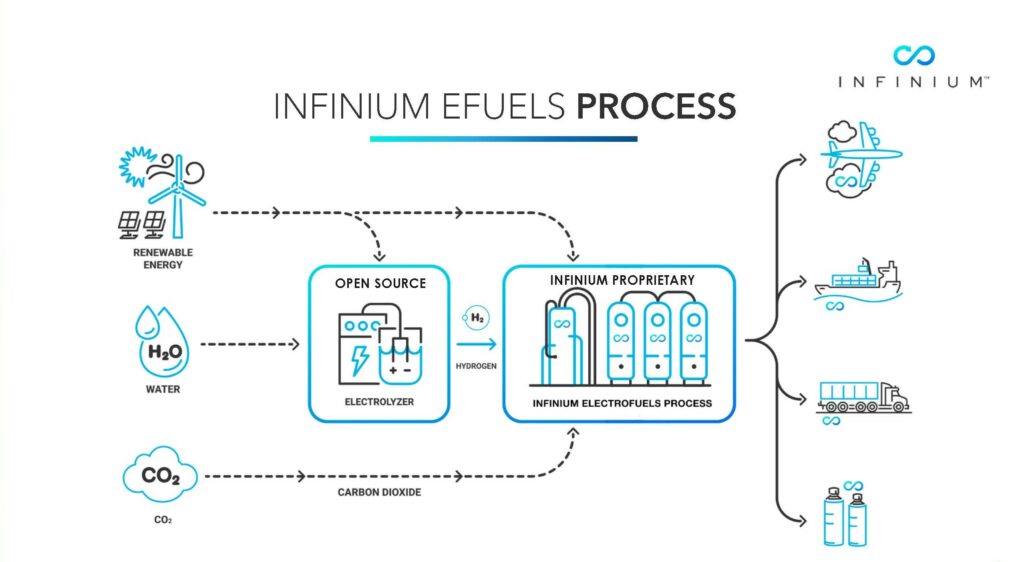
Figure 1
ENGIE, France:
Infinium have announced a fuel REUZE facility which will use 300,000 tonnes per year CO2 which will produce 100,000 tonnes per year of e-fuels which is equivalent to about 2500 bpd fuel. Byproduct steam will be used by Arcelor Mittal steel plant and oxygen will be supplied to nearby industries. The facility will be built in Dunkirk area. The plant is expected to be completed by year 2026 at an estimated investment of EUR 500 million.
Mo Industrial Park, Norway:
In January 2024, Infinium announced that e-fuel project will be developed in Mo Industrial Park, Mo I Rana, North Norway. The capacity of the plant is stated to 2000 barrels per day. The plant will produce eSAF, eDiesel and eNaphtha. No information has been disclosed on the level of investment.

